Our Company
Our Therapeutic Areas
Adverse event reporting available at the end of the page
We are proud to have four licensed medicines in the UK, offering differentiated treatments for patients with unmet needs. Below, you can find information about the disease areas we focus on, along with links to further information and guidance on reporting adverse events.
ATTR amyloidosis is a rare, progressive disease caused by the build-up of amyloid fibrils in organs and tissues, leading to various symptoms depending on the organs affected. Our work in this area focuses on providing treatments that help manage the disease.
ONPATTRO (patisiran) is indicated in the UK for the treatment of hereditary transthyretin-mediated amyloidosis (hATTR amyloidosis) in adult patients with stage 1 or stage 2 polyneuropathy.
AMVUTTRA▼(vutrisiran) is indicated in the UK for the treatment of hereditary transthyretin-mediated amyloidosis (hATTR) in adult patients with stage 1 or stage 2 polyneuropathy, as well as for wild-type or hereditary transthyretin amyloidosis in adult patients with cardiomyopathy.
Acute Hepatic Porphyria (AHP)
AHP encompasses a group of rare genetic conditions characterised by a deficiency in enzymes involved in the production of heme. This can lead to a build-up of toxic substances causing severe, potentially life-threatening attacks. AHP can cause a range of symptoms, from severe abdominal pain to neurological complications, significantly impacting quality of life. Understanding AHP is essential for early diagnosis and effective management.
GIVLAARI▼(givosiran) is indicated in the UK for the treatment of Acute Hepatic Porphyria in adults and adolescents aged 12 years and older
Primary Hyperoxaluria Type 1 (PH1)
PH1 is a rare genetic disorder resulting in the overproduction of oxalate, leading to kidney damage and other complications. Managing this condition is crucial to preventing severe health outcomes, including organ failure.
OXLUMO (lumasiran) is indicated in the UK for the treatment of Primary Hyperoxaluria Type 1 (PH1) in all age groups
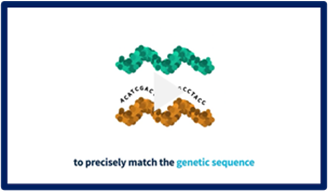
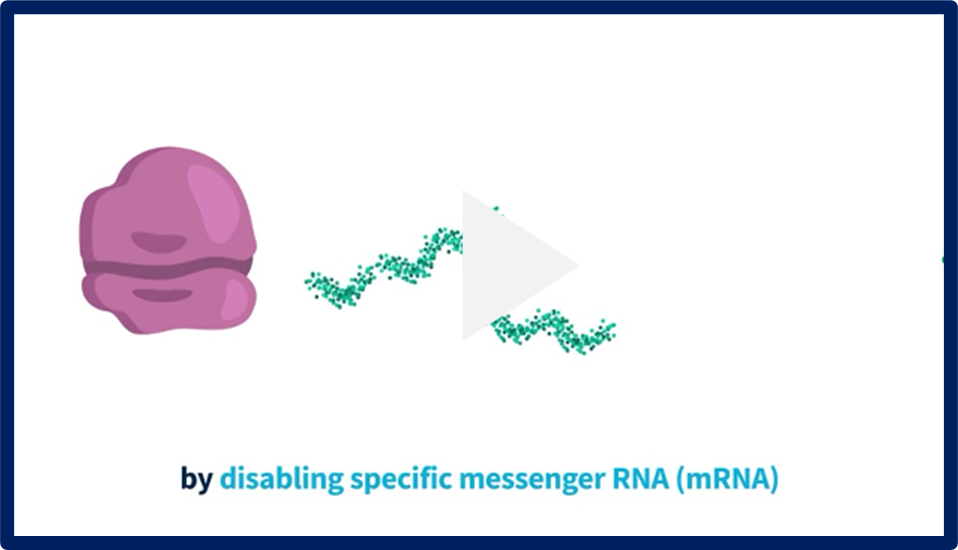
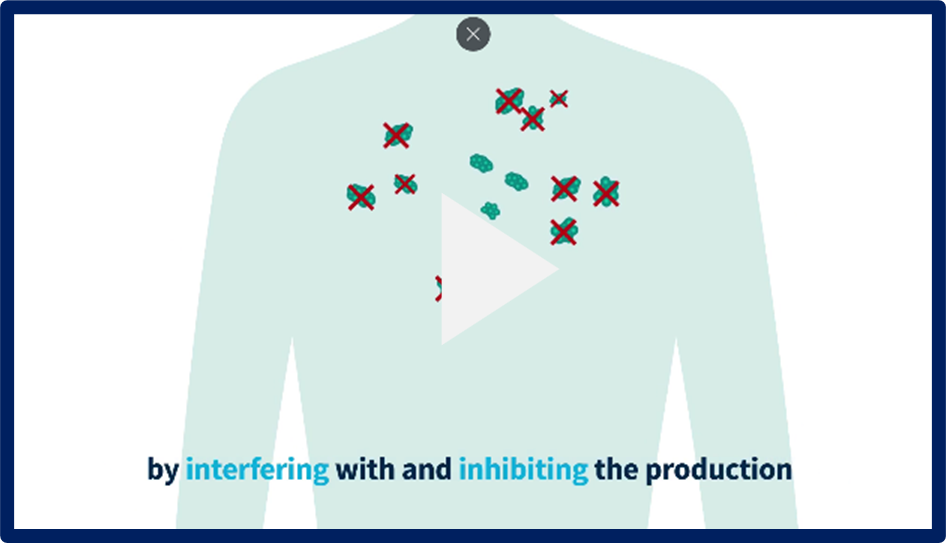
Information on RNAi Therapeutics
What are RNAi therapeutics and how do they work? Watch these educational videos to find out more.
Stay In Touch
If you’d like to stay in touch with us, please click here
Reporting of Adverse Events
Adverse events should be reported. Reporting forms and information can be found at the Medicines and Healthcare Regulatory Authority, via the Yellow Card Scheme, Website: https://yellowcard.mhra.gov.uk/ or search for MHRA Yellow Card in the Google Play or Apple App Store for the UK. Suspected adverse events can also be reported by calling the Yellow Card reporting line, for free, on 0800 731 6789.
When reporting please provide as much information as possible, including information about medical history, any concomitant medication, onset timing, treatment dates, and product brand name.
Corp-UK-00096 | February 2026